Vincent Roy1, Olivier Gagné2, Karim Hamiche3, Jean-Claude Labbé3, and Patrick Narbonne1,2§
1Département de Biologie Moléculaire, de Biochimie Médicale et de Pathologie, Faculté de Médecine, Université Laval, Québec, Canada.
2Département de Biologie Médicale, Université du Québec à Trois-Rivières, Trois-Rivières, Canada.
3Département de Pathologie et Biologie Cellulaire, Institut de Recherche en Immunologie et en Cancérologie (IRIC), Université de Montréal, Montréal, Canada.
§Correspondence to: Patrick Narbonne (Patrick.Narbonne@uqtr.ca)
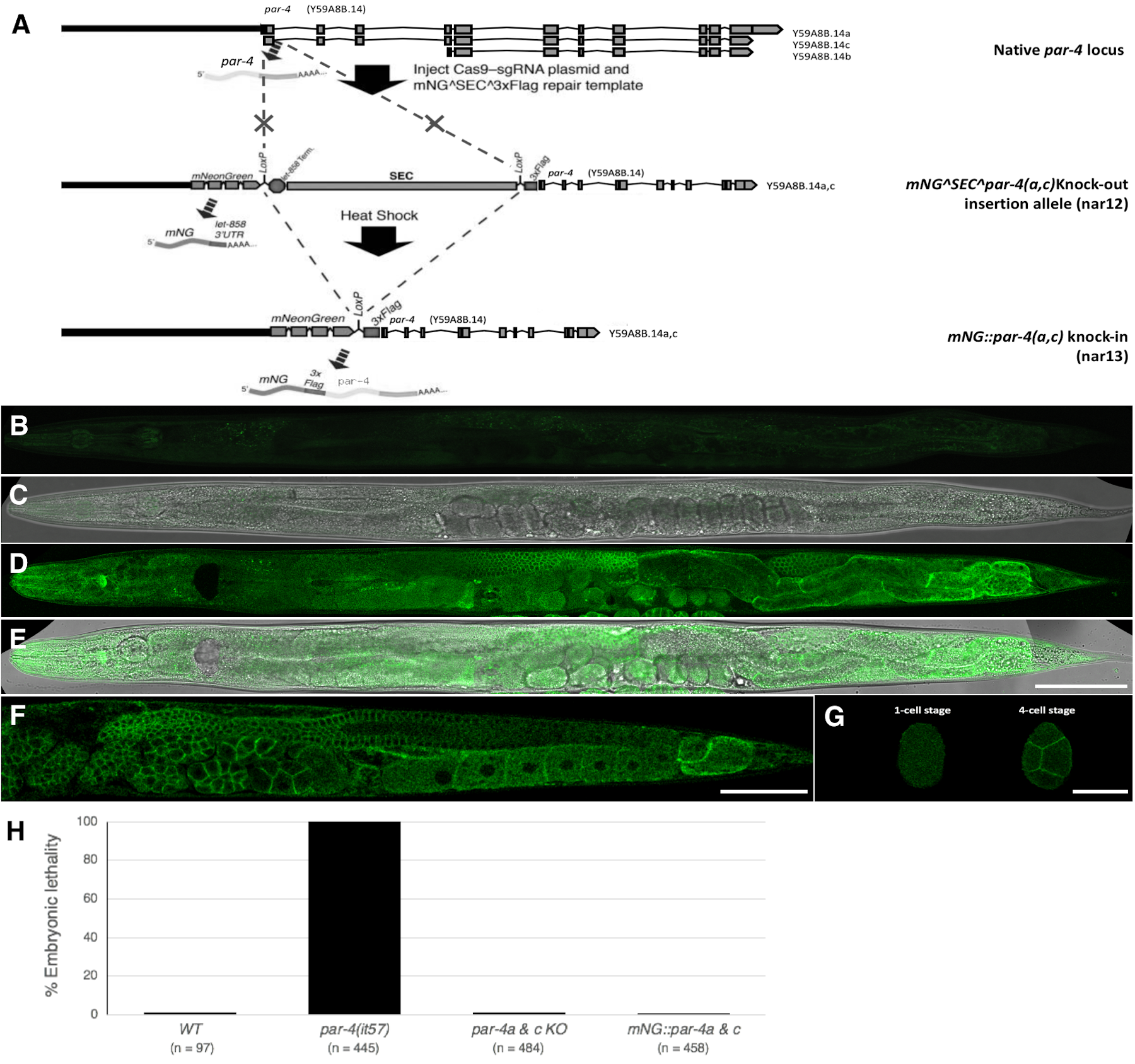
Description
Reagents
Acknowledgements
Funding
Reviewed By
Anonymous
Database Reference ID: WBPaper00055992
History
- Received: 10/11/2018
- Accepted: 11/12/2018
- Published: 11/13/2018
Copyright
© 2018 by the authors. This is an open-access article distributed under the terms of the Creative Commons Attribution 4.0 International (CC BY 4.0) License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation
PubMed Central: 7255809
PubMed: 32550374

