Nathalie Lancy Momplaisir1, Aurora Turgeon2, Jodi Flaws2, and Jing Yang2§
1Tuskegee University, Tuskegee, AL, 36088
2University of Illinois at Urbana-Champaign, Urbana-Champaign, IL, 61802
§Correspondence to: Jing Yang (yangj@illinois.edu)
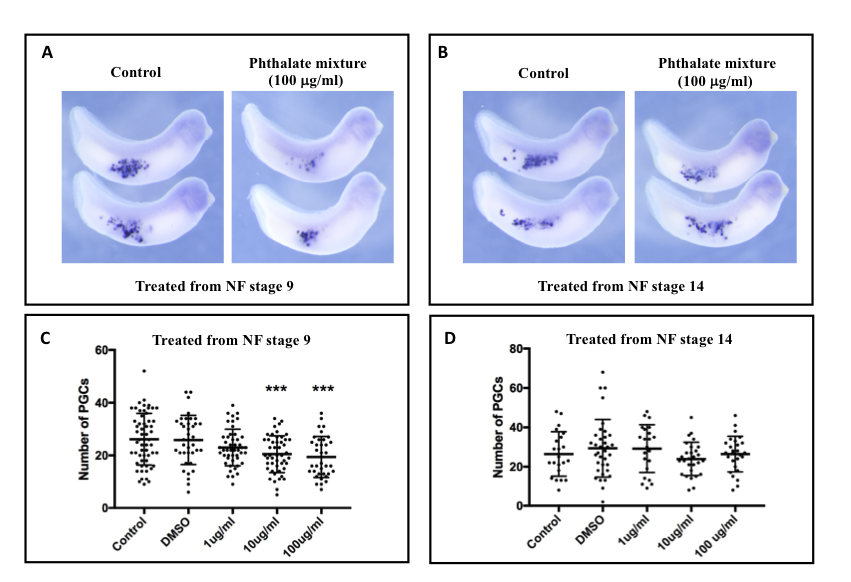
Description
Reagents
Funding
Reviewed By
Nanette Nascone-Yoder, Christina Zorn
History
- Received: 10/31/2018
- Accepted: 11/30/2018
- Published: 12/3/2018
Copyright
© 2018 by the authors. This is an open-access article distributed under the terms of the Creative Commons Attribution 4.0 International (CC BY 4.0) License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation
PubMed Central: 7255810
PubMed: 32550375
microPublication Biology:ISSN: 2578-9430

