Anna C. Erpf1,2, and Tamara Mikeladze-Dvali1§
1Department of Cell and Developmental Biology, Ludwig-Maximilians-University Munich, Grosshaderner Str. 2, 82152 Planegg-Martinsried, Germany
2Current address: Lunenfeld-Tanenbaum Research Institute, Sinai Health System, 600 University Avenue, Toronto, ON M5G 1X5, Canada
§Correspondence to: Tamara Mikeladze-Dvali (tmdvali@biologie.uni-muenchen.de)
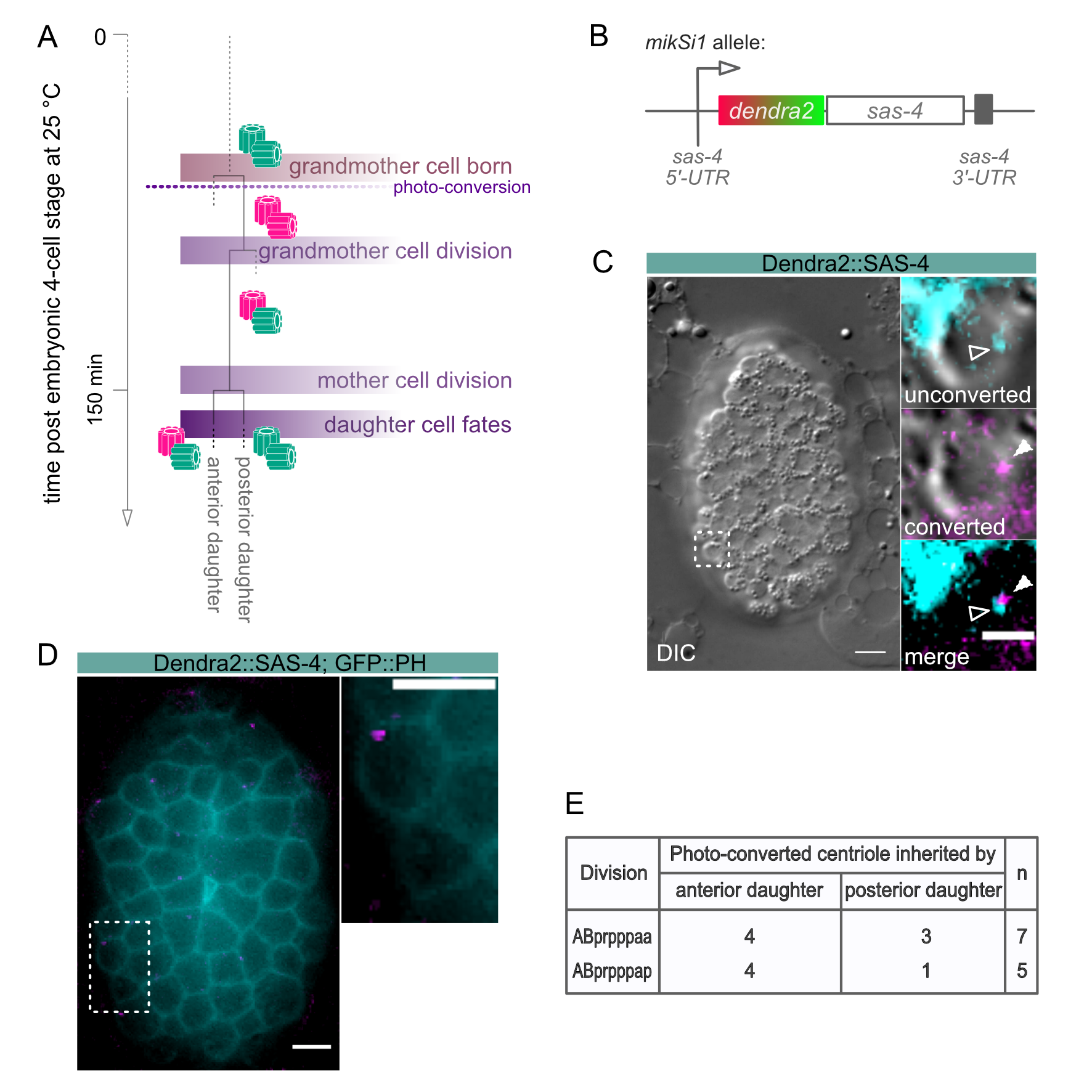
Description
Methods
Reagents
Acknowledgements
Funding
Author Contributions
- Anna C. Erpf: Conceptualization, Methodology, Writing - original draft, Writing - review and editing, Investigation
- Tamara Mikeladze-Dvali: Conceptualization, Methodology, Funding acquisition, Writing - review and editing
Reviewed By
Jyoti Iyer, Anonymous
Database Reference ID: WBPaper00059660
History
- Received: 3/12/2020
- Revision Received: 4/18/2020
- Accepted: 5/17/2020
- Published: 5/28/2020
Copyright
© 2020 by the authors. This is an open-access article distributed under the terms of the Creative Commons Attribution 4.0 International (CC BY 4.0) License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation
PubMed Central: PMC7255964
PubMed: 32550519

