Troy A McDiarmid1*, Lexis D Kepler1*, and Catharine H Rankin1,2§
1Djavad Mowafaghian Centre for Brain Health, University of British Columbia, 2211 Wesbrook Mall, Vancouver, British Columbia V6T 2B5, Canada
2Department of Psychology, University of British Columbia, 2136 West Mall, Vancouver, British Columbia V6T 1Z4, Canada
§Correspondence to: Catharine H Rankin (crankin@psych.ubc.ca)
* Authors have equal contribution
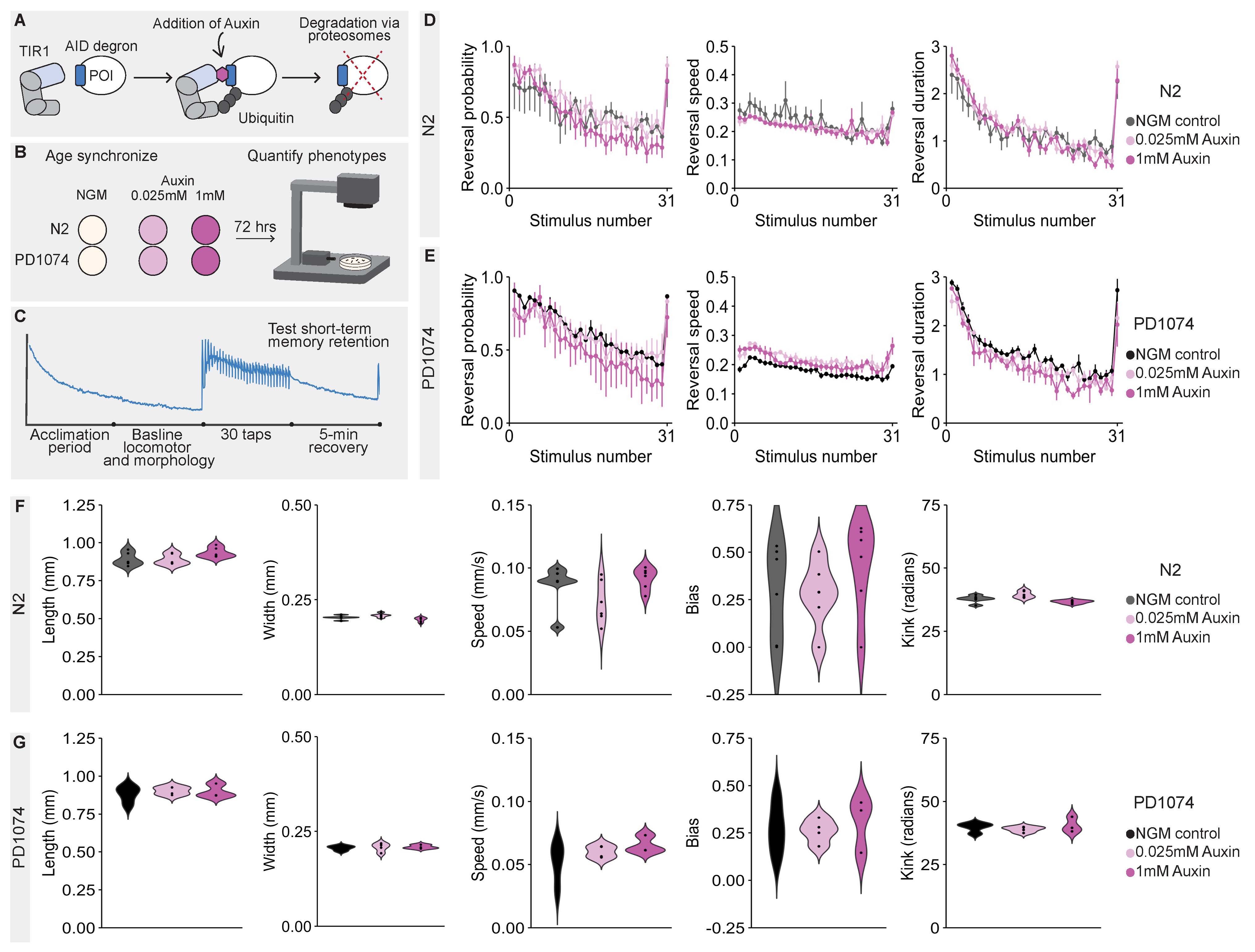
Description
Methods
Reagents
Extended Data
- Description: This repo contains Custom R scripts organized and summarized Choreography output files as well as associated data.. Resource Type: Software. DOI: 10.22002/d1.1639
Funding
Author Contributions
- Troy A McDiarmid: Conceptualization, Formal analysis, Visualization, Writing - original draft
- Lexis D Kepler: Conceptualization, Investigation, Visualization, Writing - original draft
- Catharine H Rankin: Funding acquisition, Supervision, Writing - review and editing
Reviewed By
Anonymous
Nomenclature Validated By
Anonymous
Database Reference ID: WBPaper00060382
History
- Received: 9/4/2020
- Revision Received: 9/26/2020
- Accepted: 9/30/2020
- Published: 10/8/2020
Copyright
© 2020 by the authors. This is an open-access article distributed under the terms of the Creative Commons Attribution 4.0 International (CC BY 4.0) License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation
PubMed Central: PMC7723422
PubMed: 33313486

