Woojin Kang1§, Kenji Yamatoya2, Kenji Miyado1, Mami Miyado3, and Yoshitaka Miyamoto1
1Department of Reproductive Biology, National Research Institute for Child Health and Development, Setagaya, Tokyo 157-8535, Japan
2Institute for Environmental and Gender-Specific Medicine, Juntendo University Graduate School of Medicine, Urayasu, Chiba 279-0021, Japan
3Department of Molecular Endocrinology, National Research Institute for Child Health and Development, Setagaya, Tokyo 157-8535, Japan
§Correspondence to: Woojin Kang (kwjbear@gmail.com)
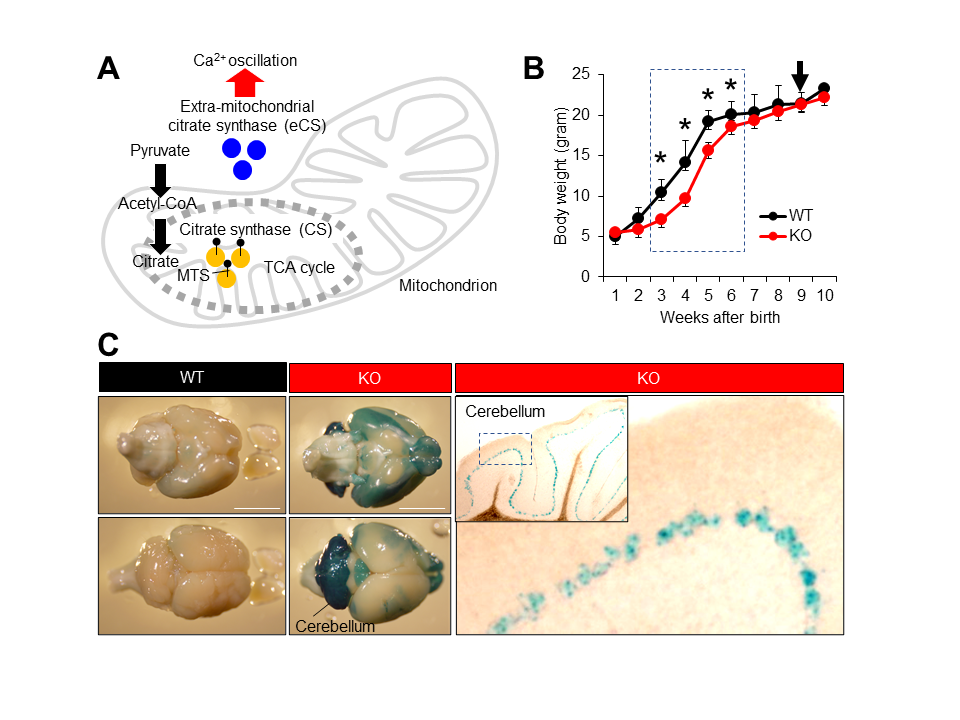
Description
Methods
Acknowledgements
Funding
Author Contributions
- Woojin Kang: Supervision, Writing - review and editing
- Kenji Yamatoya: Investigation
- Kenji Miyado: Writing - original draft, Writing - review and editing
- Mami Miyado: Investigation
- Yoshitaka Miyamoto: Investigation
Reviewed By
Anonymous
Nomenclature Validated By
Anonymous
History
- Received: 10/8/2020
- Revision Received: 11/10/2020
- Accepted: 11/10/2020
- Published: 11/12/2020
Copyright
© 2020 by the authors. This is an open-access article distributed under the terms of the Creative Commons Attribution 4.0 International (CC BY 4.0) License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation
PubMed Central: PMC7704253
PubMed: 33274323

