Hiroshi Qadota1, Yating Luo1, Andres F Oberhauser2, and Guy M Benian1§
1Department of Pathology, Emory University, Atlanta, Georgia
2Department of Neuroscience, Cell Biology & Anatomy, Sealy Center for Structural Biology and Molecular Biophysics, University of Texas Medical Branch, Galveston, Texas
§Correspondence to: Guy M Benian (pathgb@emory.edu)
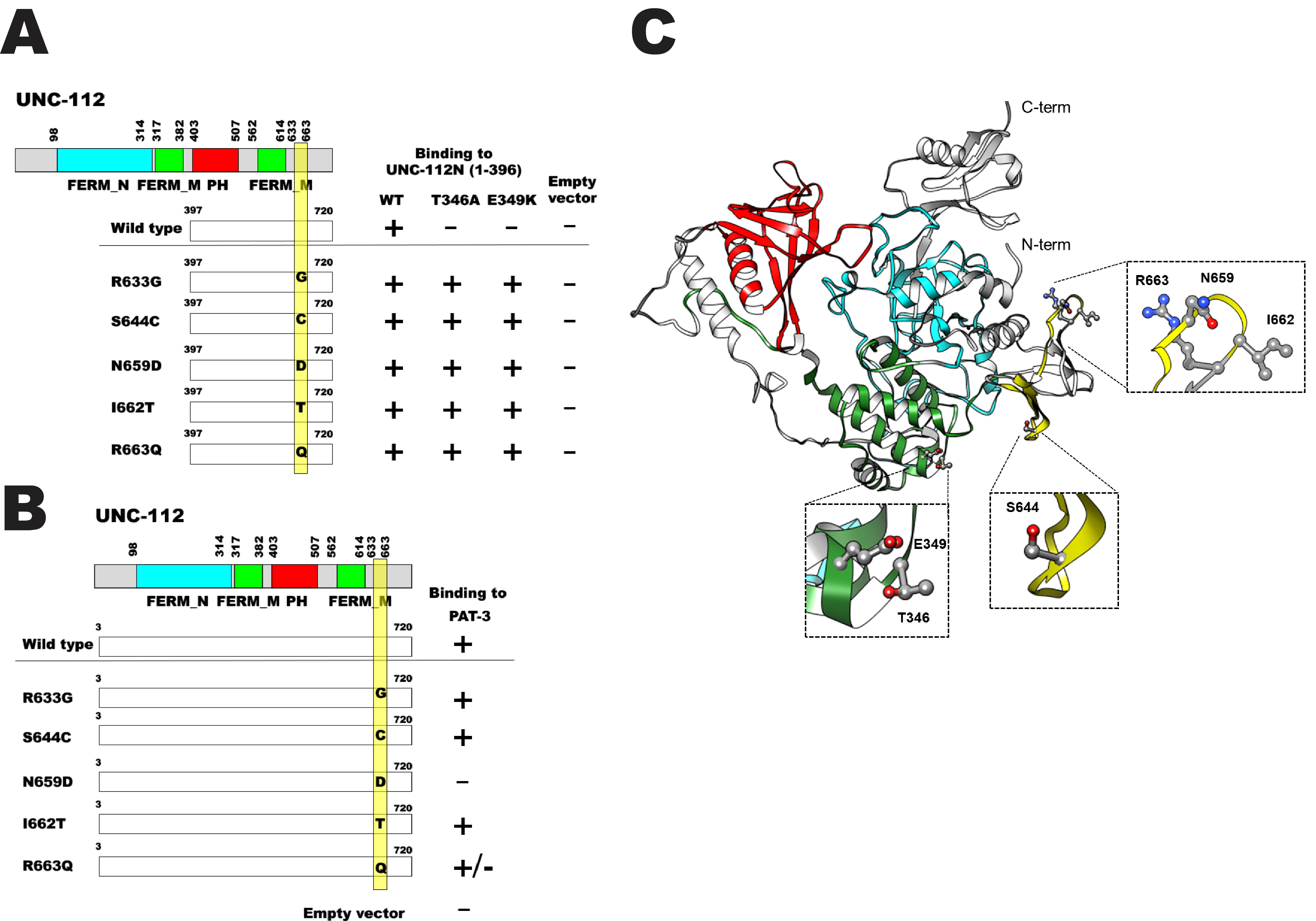
Description
Methods
Funding
Author Contributions
- Hiroshi Qadota: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Supervision, Validation, Visualization, Writing - original draft
- Yating Luo: Data curation, Investigation, Methodology
- Andres F Oberhauser: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Software, Visualization, Writing - original draft, Writing - review and editing
- Guy M Benian: Conceptualization, Data curation, Funding acquisition, Project administration, Resources, Supervision, Writing - original draft, Writing - review and editing
Reviewed By
Anonymous
Nomenclature Validated By
Chris Grove
Database Reference ID: WBPaper00060776
History
- Received: 12/2/2020
- Revision Received: 12/11/2020
- Accepted: 12/16/2020
- Published: 12/17/2020
Copyright
© 2020 by the authors. This is an open-access article distributed under the terms of the Creative Commons Attribution 4.0 International (CC BY 4.0) License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation
PubMed Central: PMC7747009
PubMed: 33367222
microPublication Biology:ISSN: 2578-9430

