Elyse M Talley1*, Charlie T Watts1*, Sonia Aboyer2, Madeline G Adamson1, Harriet AB Akoto2, Haley Altemus3, Philip J Avella2, Rebecca Bailey1, Elizabeth R Bell1, Katheryn L Bell1, Kelsey Breneman1, Jessica S Burkhart2, Logan J Chanley1, Savannah S Cook1, Mackenzie T DesLaurier2, Timothy R Dorsey2, Cassandra J Doyle1, Merris E Egloff1, Ayoola S Fasawe2, Katy K Garcia3, Nathaniel P Graves3, Tyler K Gray2, Evan M Gustafson2, Makayla J Hall2, Jaden D Hayes1, Lindsay J Holic2, Brice A Jarvis2, Piotr S Klos2, Sidney Kritzmire1, Lera Kuzovko2, Edwyna Lainez3, Shamerra McCoy3, James C Mierendorf2, Nicole A Neri2, Caley R Neville3, Kelley Osborn2, Kaitlyn Parker3, Megan E Parks2, Kylee Peck1, Robyn Pitt2, Matthew E Platta2, Brianna Powell3, Katalina Rodriguez1, Clara Ruiz2, Mariah N Schaefer1, Amanda B Shields1, Jasmine B Smiley3, Briona Stauffer3, Devan Straub1, John L Sweeney3, Kaitlyn M Termine2, Brett Thomas3, Sophia D Toth1, Taylor R Veile2, Kayla S Walker3, Paige N Webster1, Brian J Woodard1, Quentin L Yoder1, McKenzie K Young1, McKenzie L Zeedyk2, Logan N Ziegler2, Kayla L Bieser4, David P Puthoff3, Joyce Stamm1, Alysia D Vrailas-Mortimer2, Jacob D Kagey5§, and Julie A Merkle1
1University of Evansville, Evansville, IN USA
2Illinois State University, Normal, IL USA
3Frostburg State University, Frostburg, MD USA
4Nevada State College, Henderson, NV USA
5University of Detroit Mercy, Detroit, MI USA
§Correspondence to: Jacob D Kagey (kageyja@udmercy.edu)
* Authors have equal contribution
Abstract
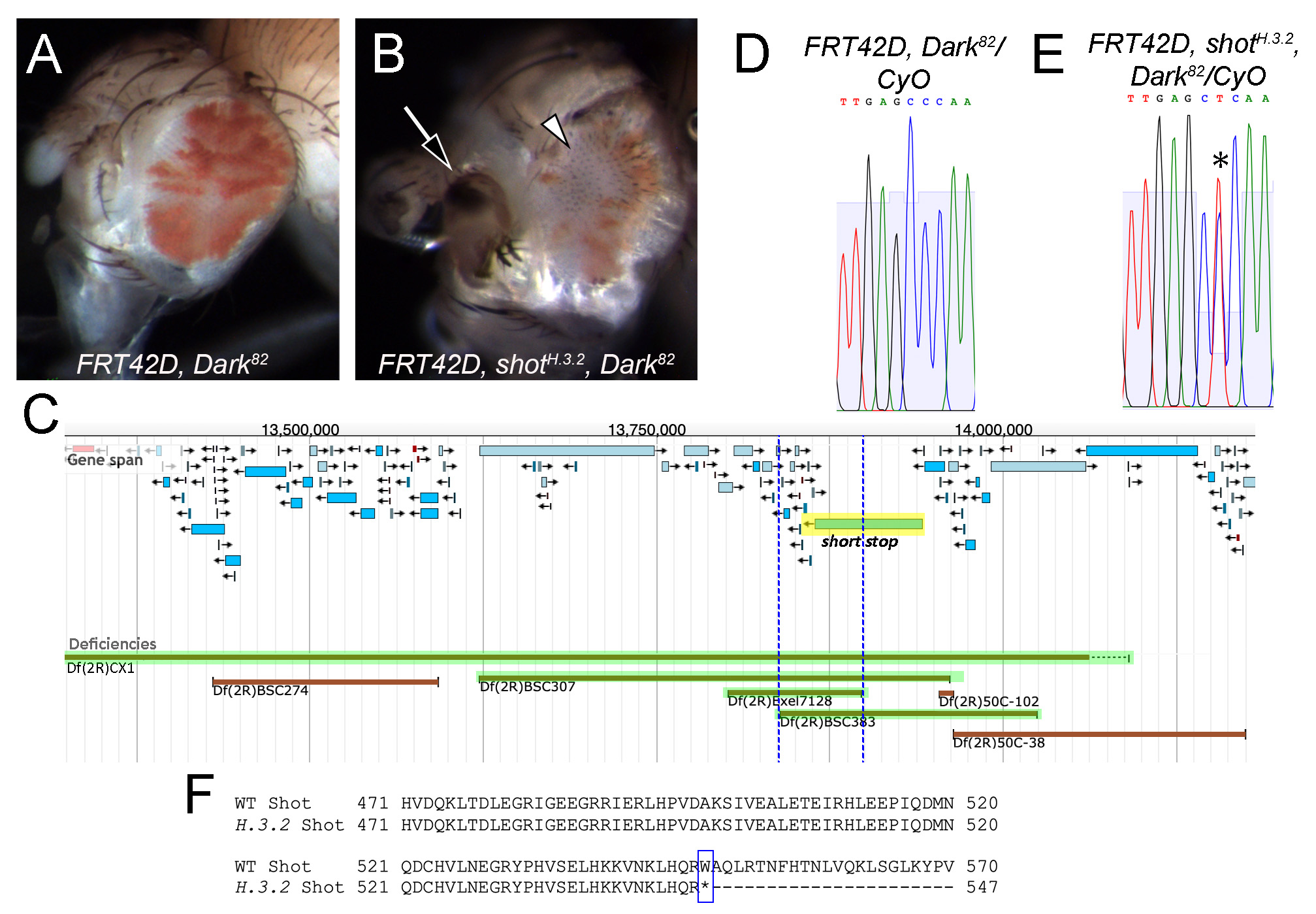
Description
Reagents
Acknowledgements
Funding
Author Contributions
- Elyse M Talley: Data curation, Formal analysis, Writing - original draft, Investigation, Writing - review and editing
- Charlie T Watts: Data curation, Formal analysis, Investigation, Writing - original draft, Writing - review and editing
- Sonia Aboyer: Data curation, Formal analysis, Investigation, Writing - review and editing
- Madeline G Adamson: Data curation, Investigation, Formal analysis, Writing - review and editing
- Harriet AB Akoto: Data curation, Investigation, Formal analysis, Writing - review and editing
- Haley Altemus: Data curation, Investigation, Formal analysis, Writing - review and editing
- Philip J Avella: Data curation, Investigation, Formal analysis, Writing - review and editing
- Rebecca Bailey: Data curation, Investigation, Formal analysis, Writing - review and editing
- Elizabeth R Bell: Data curation, Investigation, Formal analysis, Writing - review and editing
- Katheryn L Bell: Data curation, Investigation, Formal analysis, Writing - review and editing
- Kelsey Breneman: Data curation, Investigation, Formal analysis, Writing - review and editing
- Jessica S Burkhart: Data curation, Investigation, Formal analysis, Writing - review and editing
- Logan J Chanley: Data curation, Investigation, Formal analysis, Writing - review and editing
- Savannah S Cook: Data curation, Investigation, Formal analysis, Writing - review and editing
- Mackenzie T DesLaurier: Data curation, Investigation, Formal analysis, Writing - review and editing
- Timothy R Dorsey: Data curation, Investigation, Formal analysis, Writing - review and editing
- Cassandra J Doyle: Data curation, Investigation, Formal analysis, Writing - review and editing
- Merris E Egloff: Data curation, Investigation, Formal analysis, Writing - review and editing
- Ayoola S Fasawe: Data curation, Investigation, Formal analysis, Writing - review and editing
- Katy K Garcia: Data curation, Investigation, Formal analysis, Writing - review and editing
- Nathaniel P Graves: Data curation, Investigation, Formal analysis, Writing - review and editing
- Tyler K Gray: Data curation, Investigation, Formal analysis, Writing - review and editing
- Evan M Gustafson: Data curation, Investigation, Formal analysis, Writing - review and editing
- Makayla J Hall: Data curation, Investigation, Formal analysis, Writing - review and editing
- Jaden D Hayes: Data curation, Investigation, Formal analysis, Writing - review and editing
- Lindsay J Holic: Data curation, Investigation, Formal analysis, Writing - review and editing
- Brice A Jarvis: Data curation, Investigation, Formal analysis, Writing - review and editing
- Piotr S Klos: Data curation, Investigation, Formal analysis, Writing - review and editing
- Sidney Kritzmire: Data curation, Investigation, Formal analysis, Writing - review and editing
- Lera Kuzovko: Data curation, Investigation, Formal analysis, Writing - review and editing
- Edwyna Lainez: Data curation, Investigation, Formal analysis, Writing - review and editing
- Shamerra McCoy: Data curation, Investigation, Formal analysis, Writing - review and editing
- James C Mierendorf: Data curation, Investigation, Formal analysis, Writing - review and editing
- Nicole A Neri: Data curation, Investigation, Formal analysis, Writing - review and editing
- Caley R Neville: Data curation, Investigation, Formal analysis, Writing - review and editing
- Kelley Osborn: Data curation, Investigation, Formal analysis, Writing - review and editing
- Kaitlyn Parker: Data curation, Investigation, Formal analysis, Writing - review and editing
- Megan E Parks: Data curation, Investigation, Formal analysis, Writing - review and editing
- Kylee Peck: Data curation, Investigation, Formal analysis, Writing - review and editing
- Robyn Pitt: Data curation, Investigation, Formal analysis, Writing - review and editing
- Matthew E Platta: Data curation, Investigation, Formal analysis, Writing - review and editing
- Brianna Powell: Data curation, Investigation, Formal analysis, Writing - review and editing
- Katalina Rodriguez: Data curation, Investigation, Formal analysis, Writing - review and editing
- Clara Ruiz: Data curation, Investigation, Formal analysis, Writing - review and editing
- Mariah N Schaefer: Data curation, Investigation, Formal analysis, Writing - review and editing
- Amanda B Shields: Data curation, Investigation, Formal analysis, Writing - review and editing
- Jasmine B Smiley: Data curation, Investigation, Formal analysis, Writing - review and editing
- Briona Stauffer: Data curation, Investigation, Formal analysis, Writing - review and editing
- Devan Straub: Data curation, Investigation, Formal analysis, Writing - review and editing
- John L Sweeney: Data curation, Investigation, Formal analysis, Writing - review and editing
- Kaitlyn M Termine: Data curation, Investigation, Formal analysis, Writing - review and editing
- Brett Thomas: Data curation, Investigation, Formal analysis, Writing - review and editing
- Sophia D Toth: Data curation, Investigation, Formal analysis, Writing - review and editing
- Taylor R Veile: Data curation, Investigation, Formal analysis, Writing - review and editing
- Kayla S Walker: Data curation, Investigation, Formal analysis, Writing - review and editing
- Paige N Webster: Data curation, Investigation, Formal analysis, Writing - review and editing
- Brian J Woodard: Data curation, Investigation, Formal analysis, Writing - review and editing
- Quentin L Yoder: Data curation, Investigation, Formal analysis, Writing - review and editing
- McKenzie K Young: Data curation, Investigation, Formal analysis, Writing - review and editing
- McKenzie L Zeedyk: Data curation, Investigation, Formal analysis, Writing - review and editing
- Logan N Ziegler: Data curation, Investigation, Formal analysis, Writing - review and editing
- Kayla L Bieser: Data curation, Investigation, Formal analysis, Writing - review and editing, Project administration, Funding acquisition, Conceptualization, Methodology
- David P Puthoff: Data curation, Investigation, Formal analysis, Writing - review and editing, Project administration, Supervision, Validation
- Joyce Stamm: Data curation, Formal analysis, Investigation, Project administration, Validation, Writing - review and editing, Supervision, Funding acquisition
- Alysia D Vrailas-Mortimer: Data curation, Formal analysis, Investigation, Project administration, Validation, Writing - review and editing, Funding acquisition, Supervision
- Jacob D Kagey: Data curation, Investigation, Writing - review and editing, Formal analysis, Project administration, Validation, Resources, Conceptualization, Funding acquisition, Methodology
- Julie A Merkle: Data curation, Investigation, Supervision, Writing - original draft, Writing - review and editing, Formal analysis, Project administration, Validation
Reviewed By
Anonymous
History
- Received: 6/22/2021
- Revision Received: 7/1/2021
- Accepted: 7/2/2021
- Published: 7/13/2021
Copyright
© 2021 by the authors. This is an open-access article distributed under the terms of the Creative Commons Attribution 4.0 International (CC BY 4.0) License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation
PubMed Central: PMC8278230
PubMed: 34278244

