Dillon E. Sloan1, and Joshua N. Bembenek2§
1University of North Carolina, Chapel Hill
2University of Michigan
§Correspondence to: Joshua N. Bembenek (bembenek@umich.edu)
Abstract
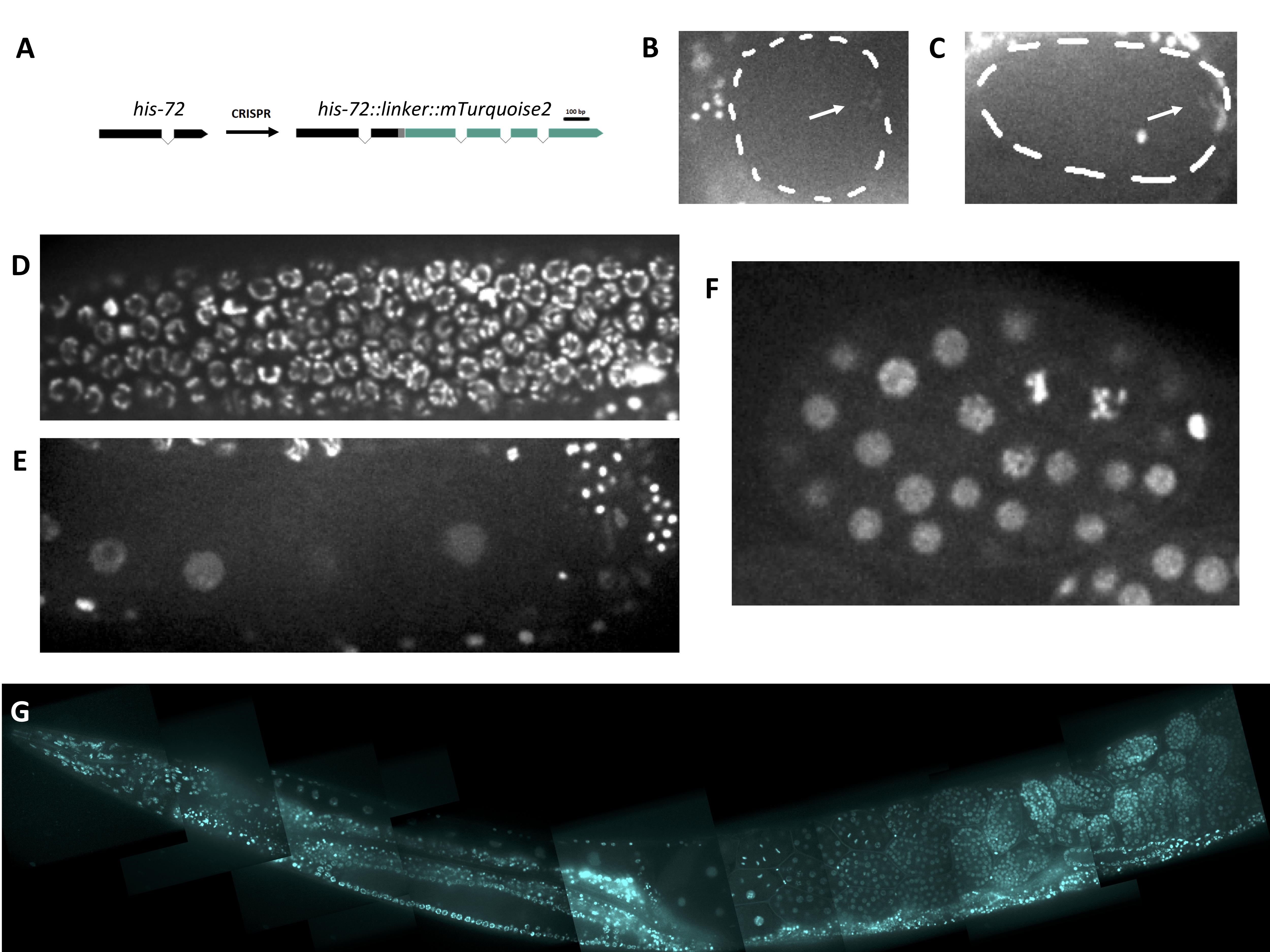
Description
Methods
Reagents
Acknowledgements
Funding
Author Contributions
- Dillon E. Sloan: Data curation, Formal analysis, Investigation, Methodology, Writing - original draft, Writing - review and editing, Software
- Joshua N. Bembenek: Conceptualization, Funding acquisition, Methodology, Project administration, Resources, Supervision, Validation, Writing - review and editing
Reviewed By
Anonymous
Nomenclature Validated By
Daniela Raciti
Database Reference ID: WBPaper00061920
History
- Received: 8/24/2021
- Revision Received: 9/11/2021
- Accepted: 9/13/2021
- Published: 9/29/2021
Copyright
© 2021 by the authors. This is an open-access article distributed under the terms of the Creative Commons Attribution 4.0 International (CC BY 4.0) License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation
PubMed Central: PMC8482033
PubMed: 34604717
microPublication Biology:ISSN: 2578-9430

