Ailén Authier1,2,3, Pablo Cerdán1,2, and Gabriela A. Auge1,2,3§
1Fundación Instituto Leloir, Buenos Aires, Argentina
2Consejo Nacional de Investigaciones Científicas y Tecnológicas (CONICET), Argentina
3Instituto de Biociencias, Biotecnología y Biología Traslacional (iB3), Facultad de Ciencias Exactas y Naturales, Universidad de Buenos Aires, Buenos Aires, Argentina
§Correspondence to: Gabriela A. Auge (gauge@fbmc.fcen.uba.ar)
This article has a correction:
Abstract
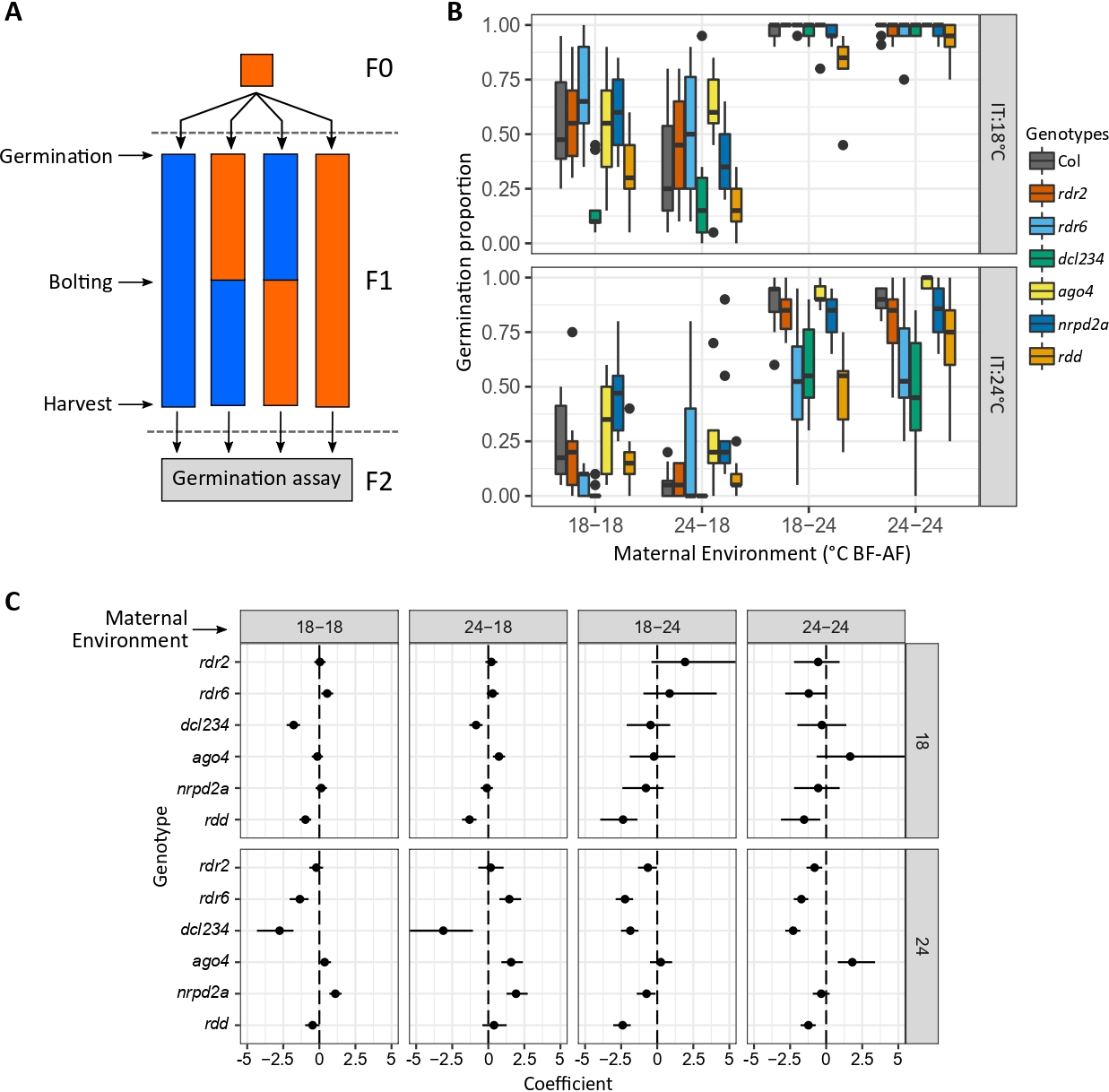
Description
Methods
Reagents
Extended Data
- Description: Raw data for the article "Role of the RNA-directed DNA Methylation pathway in the regulation of maternal effects in Arabidopsis thaliana seed germination". The file shows the final germination counts and the total number of incubated seeds for the mutant lines used in the study to test for maternal effects of ambient temperatures on seeds of Arabidopsis thaliana.. Resource Type: Dataset. DOI: 10.22002/D1.20028
Acknowledgements
Funding
Author Contributions
- Ailén Authier: Formal analysis, Investigation, Visualization, Writing - original draft, Writing - review and editing
- Pablo Cerdán: Funding acquisition, Resources, Writing - review and editing
- Gabriela A. Auge: Conceptualization, Investigation, Formal analysis, Funding acquisition, Supervision, Writing - review and editing, Visualization
Reviewed By
Anonymous
History
- Received: 9/2/2021
- Revision Received: 10/11/2021
- Accepted: 11/3/2021
- Published: 12/6/2021
Copyright
© 2021 by the authors. This is an open-access article distributed under the terms of the Creative Commons Attribution 4.0 International (CC BY 4.0) License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation
PubMed Central: PMC8652579
PubMed: 34901781

